International System of Units
 |
Contents |
The International System of Units are more commonly know as SI Units. The reason it isn’t IS is because its from the French, Le Systeme International. It is the world’s most widely used system of units, both in everyday commerce and in science. It is a beacon of ability of science to unify people like no other discipline. Regardless of nationality, race, religious views, hairstyle or favourite colour nearly every scientist in the world use SI units, with most countries not even maintaining official definitions of any other units. There are only 3 countries that havent oficially adopted SI as the primary system of measurment, Liberia, Myanmar and the USA.
The international system of units consists of a set of units together with a set of prefixes. The units describe length, time, mass, temperature, current and luminous intensity. From these 7 base units all other units are derived.
Fundamental Units
Metre (m)
Hopefully you’ll already be familiar with the metre. It’s the SI unit of length. 1m is equal to the length of the path travelled by light in a vacuum during the time interval of 1/299,792,458 of a second. All lengths in physics are given in metres (with or without SI prefixes).
Seconds (s)
I’d be a little worried if you didn’t know about this one. The Second is the SI unit of time. 1s is the duration of exactly 9,192,631,770 periods of the radiation corresponding to the transition between two hyperfine levels of the ground state of the caesium-133 atom at a temperature of 0 K. Which if you understood, you’re probably in the wrong place. A second is 1/60th of a minute, which is 1/60th of an hour, which is 1/24th of a day. 365 days in a year, blah, blah, blah. You get the picture.
Kilogram (kg)
The Kilogram is th SU unit of mass. 1kg is equal to the mass of the international prototype kilogram (a platinum-iridium cylinder) kept at the Bureau International des Poids et Mesures in Paris. The kilogram is the only base unit with a prefix, which might seem a little odd, I myself have always wondered why, but it seems a bit late to change it now. It’s not that tricky once you know it, just remember that when prefixes are used, kilogram, not grams, are the SI unit.
Kelvin (K)
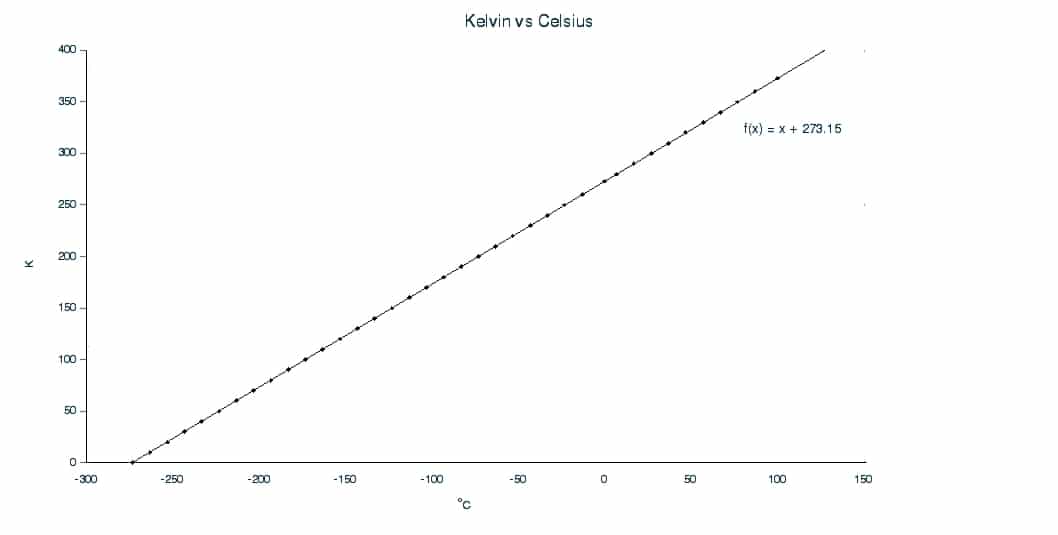
Kelvin is the SI unit of temperature. For all equations in thermodynamics, Kelvin should be used, not oC.
The graph below shows the relationship between K and oC. In order to convert from o to kelvin, simply add 273.15. 0K is also known as absolute zero, because it is the coldest temperature achievable. The Kelvin scale is also sometimes known as the Absolute Scale
Ampere (A)
The Ampere is the SI unit of electric current. 1A current is defined as the current which, if put through two straight parallel wires of infinite length which are 1 metre apart in vacuum, you would get a force between them of 2×10-7 newton per metre of length.
Mole (mol)
The mole is the SI unit of amount of substance. 1 mol is and amount of substance that contains the same number of things (atoms, molecules, ions, electrons ect) as there are atoms in 0.012 kilograms of pure carbon 12; this number is approximately equal to 6.0221415(10)x1023 mol.
Candela (cd)
The Candela is the SI unit of luminous intensity. 1cd is the luminous intensity, in a given direction, of a source that emits monochromatic radiation of frequency 540×1012 hertz and that has a radiant intensity in that direction of 1/683 watts per steradian.
SI Prefixes
The twenty SI prefixes are shown in the chart below.
| 10n | Prefix | Symbol | Decimal |
|---|---|---|---|
| 1024 | yotta | Y | 1 000 000 000 000 000 000 000 000 |
| 1021 | zetta | Z | 1 000 000 000 000 000 000 000 |
| 1018 | exa | E | 1 000 000 000 000 000 000 |
| 1015 | peta | P | 1 000 000 000 000 000 |
| 1012 | tera | T | 1 000 000 000 000 |
| 109 | giga | G | 1 000 000 000 |
| 106 | mega | M | 1 000 000 |
| 103 | kilo | k | 1 000 |
| 102 | hecto | h | 100 |
| 101 | deca | da | 10 |
| 100 | (none) | (none) | 1 |
| 10-1 | deci | d | 0.1 |
| 10-2 | centi | c | 0.01 |
| 10-3 | milli | m | 0.001 |
| 10-6 | micro | μ | 0.000 001 |
| 10-9 | nano | n | 0.000 000 001 |
| 10-12 | pico | p | 0.000 000 000 001 |
| 10-15 | femto | f | 0.000 000 000 000 001 |
| 10-18 | atto | a | 0.000 000 000 000 000 001 |
| 10-21 | zepto | z | 0.000 000 000 000 000 000 001 |
| 10-24 | yocto | y | 0.000 000 000 000 000 000 000 001 |
Fundamental Constants
Given how much in the universe is changing it may come as a bit of a surprise to know that there are many fundamental constants. These constants are, as far as we know, unchanging throughout the entire universe. Some of them are mathematical constants, some physical, all important. I won’t be listing all of them here, only some of the most important to physics.
-
0
Hopefully this one shouldn’t be new to you, however I doubt you’ve given it much thought. 0 is an even integer preceding 1. It is a natural number, a whole number, a rational number, a real number, an algebraic number, a complex number, neither positive nor negative, neither a prime number nor a composite number, nor is it a unit.
“The importance of the creation of the zero mark can never be exaggerated. No single mathematical creation has been more potent for the general on-go of intelligence and power.” G.B. Halsted
A zero result in physics can lead to new discoveries and theories. Getting a wrong answer is easy, getting zero has major repercussions.
-
1
Don’t worry, I wont be listing all of the intergers as constants, only 1 and 0, as these are the most important. 1 is unity. It is the natural number following 0 and preceding 2, and is the multiplicative identity of the integers and the real and complex numbers. Its power and simplicity can be shown in the equation 1 + 1= 2
“For this equation to come into being there had to be the invention of a method for representing a physical reality, quantities had to be given names and symbols. There had to be a system to show how these quantities could be grouped together or taken apart. The writing down of this equation gave us the ability to present ideas, to discuss concepts, which led to an ever-expanding sphere of knowledge.” Neil Blackie
-
π
The ratio of circumference to diameter of every circle in existence, C=πd. The value of Pi is infinitely long and never repeats, so cannot be expressed as a fraction or finite algebraic sum. The fact that it goes on to infinity means it contains every grouping of numbers imaginable an infinite amount of times, this is partially demonstrated by the Feynman Point.
-
e
There are 2 constants represented by e, but you can usually tell which one you’re working with. e can stand for the unit of elementary charge, or the base of the natural logarithms.
- e is the electric charge carried by a single proton or electron (althoug the signs for them are reversed, protons have +e and electrons have -e). It’s a fundamental physical constant and is classed as a natural unit. It’s roughly equal to 1.6×10-19C. It was thought that the elementary charge was indivisible, however Quarks were found to have fractional electric charges in units of 1/3 e and 2/3 e so that now the term elementary charge referring to the charge on an electron is no longer strictly correct. However since quarks are not detected except in groupings that have charges that are integer multiples of e it remains a fundamental constant.
- e is the value of one of the most important mathematicl constants. It’s the base of natural logarithms. Now this may sound confusing but remember its just a number. To work out e all you have to do is put in bigger and bigger numbers for n into the following equation
(1)

The bigger the number you put in for n the closer you get to the actual value for e.
-
c
c is used to denote the speed of light in a vacuum. The reason c is used is because of the latin word for swift “Celeritas”. The value of c is exactly 299,792,458 metres per second, although a lot of the time it is rounded to 3×108ms-1